2023 HSC Chemistry Answers
The 2023 HSC Chemistry exam paper can be found on the NESA website.
Section I: Multiple-choice Questions
A and B are incorrect as hydrocarbons are toxic chemicals and should not be poured down the sink. C is incorrect as hydrocarbons are flammable and exposing it to an open flame from a match can lead to fire. Hydrocarbons need to be placed in a separate waste container labelled ‘Organic Waste’. The answer is D.
Light from a lamp is being passed through a sample. This is either colourimetry, ultraviolet-visible spectrophotometry or atomic absorption spectroscopy. Atomic absorption spectroscopy is the only technique out of the three that atomises the sample in a flame. In colourimetry and ultraviolet-visible spectrophotometry, the sample is kept in an aqueous solution form. The answer is C.
There are 5 carbons in a chain so the prefix is ‘pent’. There is a carbon-carbon triple bond, indicating an alkyne so the suffix is ‘yne’. Numbering from right to left gives the smallest location numbers for the two carbons in the triple bond (2 and 3). Take the smaller number to include in the name. The IUPAC name is 2-pentyne or pent-2-yne. The answer is B.
Write the equilibrium expression and substitute in the given concentrations.
Kₛₚ = [Na⁺][Cl⁻] = 6.13 x 6.13 = 37.6 (3 s.f.)
The answer is D.
A concentrated solution has relatively more moles in a volume than a dilute solution. A and B are incorrect as it contains less moles. A weak acid dissociates partially so there is a mixture of molecules and dissociated ions in the solution. The answer is D.
pH = -log₁₀[H⁺] so [H⁺] = 10⁻ᵖᴴ. At pH = 8, [H⁺] = 10⁻⁸ and
at pH = 5, [H⁺] = 10⁻⁵. The concentration of hydrogen ions increased by a factor of 1000. The answer is C.
The pH decreases over time so acid (with pH < 7) is being added to a base. The pH at the equivalence point is 5, indicating an acidic salt. An acidic salt is produced by titrating a strong acid with a weak base. Strong acid must be being added to a weak base. The answer is C.
A compound has a higher boiling point if it forms stronger or more intermolecular forces that require more energy to break and transition it from the liquid to the gaseous base. Heptane is a non-polar molecule that only forms weak-dispersion forces so it has the lowest boiling point. Heptan-2-one contains a C=O bond that makes it polar and allows it to form dipole-dipole forces. Heptan-1-ol can form even stronger hydrogen bonding forces and heptanoic acid can form more hydrogen bonding forces than heptan-1-ol. The answer is A.
H₂SO₄ is diprotic so [H⁺] = 2 x [H₂SO₄] = 2 x 10⁻⁵ M.
pH = -log₁₀[H⁺] = -log₁₀(2 x 10⁻⁵) = 4.7 which has a purple colour.
NaOH contains one hydroxide ion so [OH⁻] = [NaOH] = 5 x 10⁻⁵ M. pOH = -log₁₀[OH⁻] = -log₁₀(5 x 10⁻⁵) = 4.3 and
pH = 14 - pOH = 14 - 4.3 = 9.7 which has a blue-green colour.
The answer is C.
Maximising the yield of ammonia involves shifting the position of equilibrium to the right. The right hand side has less moles of gas than the left, allowing the system to decrease pressure. To influence the system to do this, a high pressure should be set. The answer is B.
All salts containing acetate ions are soluble so the answer cannot be B. Barium does not form a precipitate with bromide ions so the answer cannot be C. Magnesium does not form a precipitate with chloride ions so the answer cannot be D. The answer is A.
2HCl(aq) + Na₂CO₃(aq) → 2NaCl(aq) + CO₂(g) + H₂O(l)
MM(Na₂CO₃) = 22.99 x 2 + 12.01 + 16.00 x 3 = 105.99 g/mol
n(Na₂CO₃) = m/MM = 1.34 g/105.99 g/mol = 0.0126 mol
n(HCl) = 2 x n(Na₂CO₃) = 2 x 0.0126 = 0.0252 mol
V(HCl) = n/C = 0.0252 mol/0.54 mol/L = 0.0468 L = 46.8 mL
The answer is D.
Methanol has 1 carbon, ethanol has 2 and butan-1-ol has 4. The table shows that increasing the length of the carbon chain increases the heat of combustion. The heat of combustion of propan-1-ol with 3 carbons must be between the heat of combustions for ethanol and butan-1-ol. It can be approximated by averaging the values of the two data points between it. The answer is B.
To quantify the amount of each ion present in the mixture, the two ions need to be precipitated out at different times so it can be separated and weighed. The answer cannot be C or D. From the HSC data sheet, Agl has a smaller solubility constant (Kₛₚ) than AgCI and would precipitate first. The answer is B.
PbI₂(s) ⇌ Pb²⁺(aq) + 2I⁻(aq)
Kₛₚ = [Pb²⁺][I⁻]² = (x)(2x)² = 4x³ = 9.8 x 10⁻⁹
Molar solubility = x = 1.3 x 10⁻³ mol/L
n(PbI₂) = C x V = 1.3 x 10⁻³ mol/L x 0.375 L = 5.1 x 10⁻⁴ mol
m(PbI₂) = n x MM = 5.1 x 10⁻⁴ mol x 461 g/mol = 0.233 g
The answer is A.
At time t, both the forward and reverse reaction rates decreased. According to collision theory, the temperature must have decreased. The endothermic reaction is always more responsive to changes in heat content than exothermic reactions. The graph shows that the bolded line that decreased more is the forward reaction. The forward reaction must be endothermic and have a positive enthalpy sign (+). The answer is A.
CH₃CO has a molar mass of 43 g/mol which corresponds to the peak at m/z 43. The detector can only detect a fragment with charge. The answer is B.
Section II: Long Response Questions
Functional group isomers have different functional groups. There are two pairs of functional group isomers that could be named: butan-2-one and butanal or butan-2-one and 2-methylpropanal. Ketones and aldehydes are considered functional group isomers as ketones have a C=O bond with a non-terminal carbon and aldehydes have a C=O bond with a terminal carbon.
Chain isomers have a different arrangement of the carbon chain. There is only one pair of chain isomers: butanal and 2-methylpropanal. The position of the C=O functional group has stayed the same and only the rest of the carbon chain has changed structure.
An Arrhenius acid is a substance that produces hydrogen ions in aqueous solution. HCl is an Arrhenius acid in water because it can produce a hydrogen ion.
HCl(aq) → H⁺(aq) + Cl⁻(aq)
NH₄Cl dissolves in water to produce NH₄⁺(aq) and Cl⁻(aq). NH₄⁺ is an Arrhenius acid because it can also produce a hydrogen ion in water. NH₄Cl itself is a salt, not an Arrhenius acid.
NH₄⁺(aq) → H⁺(aq) + NH₃(aq)
A Bronsted-Lowry acid is a substance that can donate a proton to a base. HCl and NH₄⁺ are both Bronsted-Lowry acids. For instance, it can donate a proton to water in solution to produce hydronium ions. Once again, NH₄Cl itself is a salt, not a Bronsted-Lowry acid.
HCl(aq) + H₂O(l) → Cl⁻(aq) + H₃O⁺(aq)
NH₄⁺(aq) + H₂O(l) ⇌ NH₃(aq) + H₃O⁺(aq)
The pH of solution X increased from 7.00 to 12.00. The change is drastic so solution X must be water as it was initially at a neutral pH. The pH increased due to the presence of hydroxide ions from concentrated NaOH.
The pH of solution Y did not increase significantly from 7.00 to 7.02. Solution Y must be a buffer that shifted its position of equilibrium to counteract the initial increase in concentration of hydroxide ions from NaOH. The buffer could resist an increase in pH.
Amphiprotic means it can donate a proton to act as a Bronsted-Lowry acid (Equation 1) and accept a proton to act as a Bronsted-Lowry base (Equation 2).
Equation 1: HC₂O₄⁻(aq) + H₂O(l) ⇌ C₂O₄²⁻(aq) + H₃O⁺(aq)
Equation 2: HC₂O₄⁻(aq) + H₂O(l) ⇌ H₂C₂O₄(aq) + OH⁻(aq)
q = mc∆T = 205 g x 4.18 J/g/K x (60.4 - 23.7) K = 31448 J
n = -q/∆H = -31.448 kJ/-5294 kJ/mol = 0.0059 mol
m(octan-1-ol) = n x MM = 0.0059 mol x 130.23 g/mol = 0.774 g (3 s.f.)
Biofuels are more sustainable as they are renewable, meaning it is produced at an equal or faster rate than it is consumed, allowing supply to be sustained in the long term. In contrast, fossil fuels are consumed quickly and take millions of years to reform.
NO₂ and N₂O₄ exist in an equilibrium according to the following equation.
NO₂(g) ⇌ N₂O₄(g)
As NO₂ is consumed by the reaction with water, the concentration of NO₂ in the equilibrium with N₂O₄ decreases. According to Le Chatelier’s Principle, the position of equilibrium will shift to the left to produce more NO₂ to counteract the change. This allows more NO₂ to react with water in Reactor 3. The cycle continues until the supply of N₂O₄ is depleted.
Separator 1 removes and disposes of the water. Water is required as a reactant in Reactor 3. One improvement is to reuse the same water in Reactor 3 instead of disposing it. This will reduce production and disposal costs.
Heat released from the cooler/condenser can be redirected to emissions control to reduce energy costs.
m(ethanol) = 0.789 g/mL x 185 mL = 145.965 g
n(ethanol) = m/MM = 145.965 g/46.068 g/mol = 3.17 mol
n(CO₂) = n(ethanol) = 3.17 mol
P: 100 kPa = 100 000 Pa
PV = nRT
V = nRT/P = 3.17 mol x 8.314 J/mol/K x 310 K/100 000 Pa
V = 0.817 m³ = 81.7 L (3 s.f.)
The bromine water test could be done. Place a few drops of brown bromine water in a test tube with Q and mix. If bromine decolourises, it has performed a bromination reaction with the alkene to produce a dibromoalkane. This would not happen if it was not an alkene.
Take the peak at m/z = 112 as the molecular ion peak corresponding to the molar mass of the compound R with the chlorine-35 isotope. The peak at m/z = 114 is due to the less common chlorine-37 isotope.
m(C) = 112 x 32% = 35.85 ≈ 36
N(C) = 36/12 = 3
There are 3 carbons in Compound R which is an alkane produced by the addition of a diatomic chlorine molecule to an alkene Q. Compound R must be dichloropropane.
The graph shows the solubility of alkan-1-ols in water decreases as molar mass increases. Alkan-1-ol molecules consists of a polar hydroxyl (C-OH) functional group and a non-polar carbon chain. As the length of the carbon chain increases, the effect of the non-polar chain outweighs the effect of the polar hydroxyl group. The overall molecule becomes increasingly non-polar, forming more weak dispersion forces and relatively less hydrogen bonding forces needed to attract water. Therefore, solubility decreases to approximately 0 at a molar mass of 130 g/mol.
Add a few drops of calcium chloride solution to the sample and shake. Calcium can form a precipitate with carbonate ions only so a precipitate forming confirms the presence of carbonate ions.
Ca²⁺(aq) + CO₃²⁻(aq) → CaCO₃(s)
If a precipitate does form, separate it from the remaining solution before proceeding. Then, add a few drops of silver nitrate solution into the sample. If a silver bromide precipitate forms, that confirms the identity of the bromide ions.
Ag⁺(aq) + Br⁻(aq) → AgBr(s)
V(HCl) = (22.05 + 22.00 + 21.95)/3 = 22.00 mL
n(HCl) = C x V = 0.1102 mol/L x 0.022 L = 2.42 x 10⁻³ mol
HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
n(NaOH) in 20 mL aliquot = n(HCl) = 2.42 x 10⁻³ mol
n(NaOH) in 250 mL = 2.42 x 10⁻³ x 250/20 = 3.0 x 10⁻² mol
n(NaOH) added to NH₄⁺ = C x V = 1.124 mol/L x 0.05 L = 5.6 x 10⁻² mol
n(NaOH) reacted with NH₄⁺ = 5.6 x 10⁻² - 3.0 x 10⁻² = 2.6 x 10⁻² mol
n(NH₄⁺) = n(NaOH) = 2.6 x 10⁻² mol
MM(NH₄⁺) = 14.01 + 1.008 x 4 = 18.042 g/mol
m(NH₄⁺) = n x MM = 2.6 x 10⁻² mol x 18.042 g/mol = 0.4669 g (4 s.f.)
Between 6 and 8 minutes, the amounts of all substances are constant. This indicates the forward and reverse reactions are equal, giving the appearance of no macroscopic changes in the amounts. The system must be in a state of dynamic equilibrium.
One factor is an increase in volume and decrease in pressure that caused the position of equilibrium to shift to the left where there are more moles of A₂ and B₂ gases compared to AB₂ gases on the right. This allows for an increase in pressure that counteracts the disturbance.
Another factor is an increase in temperature. The system counteracts this by shifting to the left to favour the endothermic reaction that absorbs heat and decreases the temperature again.
Mg(NO₃)₂(aq) + 2NaF(aq) → MgF₂(s) + 2NaNO₃(aq)
n(NaF) added = C x V = 1.50 mol/L x 0.175 L = 0.2625 mol
n(MgF₂) produced = m/MM = 0.6231 g/62.31 g/mol = 0.01 mol
n(NaF) reacted = 2 x n(MgF₂) = 2 x 0.01 mol = 0.02 mol
n(F⁻) remaining = n(NaF) = 0.2625 - 0.02 = 0.2425 mol
[F⁻] = n/V = 0.2425 mol/(0.125 + 0.175) L = 0.8083 mol/L
MgF₂(s) ⇌ Mg²⁺(aq) + 2F⁻(aq)
Kₛₚ = [Mg²⁺][F⁻]² = (x)(0.8083 + 2x)² ≈ (x)(0.8083)² = 5.16 x 10⁻¹¹
[Mg²⁺] = x = 5.16 x 10⁻¹¹/(0.8083)² = 7.90 x 10⁻¹¹ mol/L (3 s.f.)
CHCl₂COOH(aq) + H₂O(l) ⇌ CHCl₂COO⁻(aq) + H₃O⁺(aq)
[CHCl₂COOH]ᵢ = 0.2000 mol/L
[CHCl₂COO⁻]ₑ = [H₃O⁺]ₑ = 10⁻ᵖᴴ = 10⁻¹˙¹⁰⁷ mol/L
[CHCl₂COOH]ₑ = (0.2000 - 10⁻¹˙¹⁰⁷) mol/L
Kₐ = [CHCl₂COO⁻]ₑ[H₃O⁺]ₑ/[CHCl₂COOH]ₑ
Kₐ = (10⁻¹˙¹⁰⁷)(10-¹˙¹⁰⁷)/(0.2000 - 10⁻¹˙¹⁰⁷) = 0.05014 (4 s.f.)
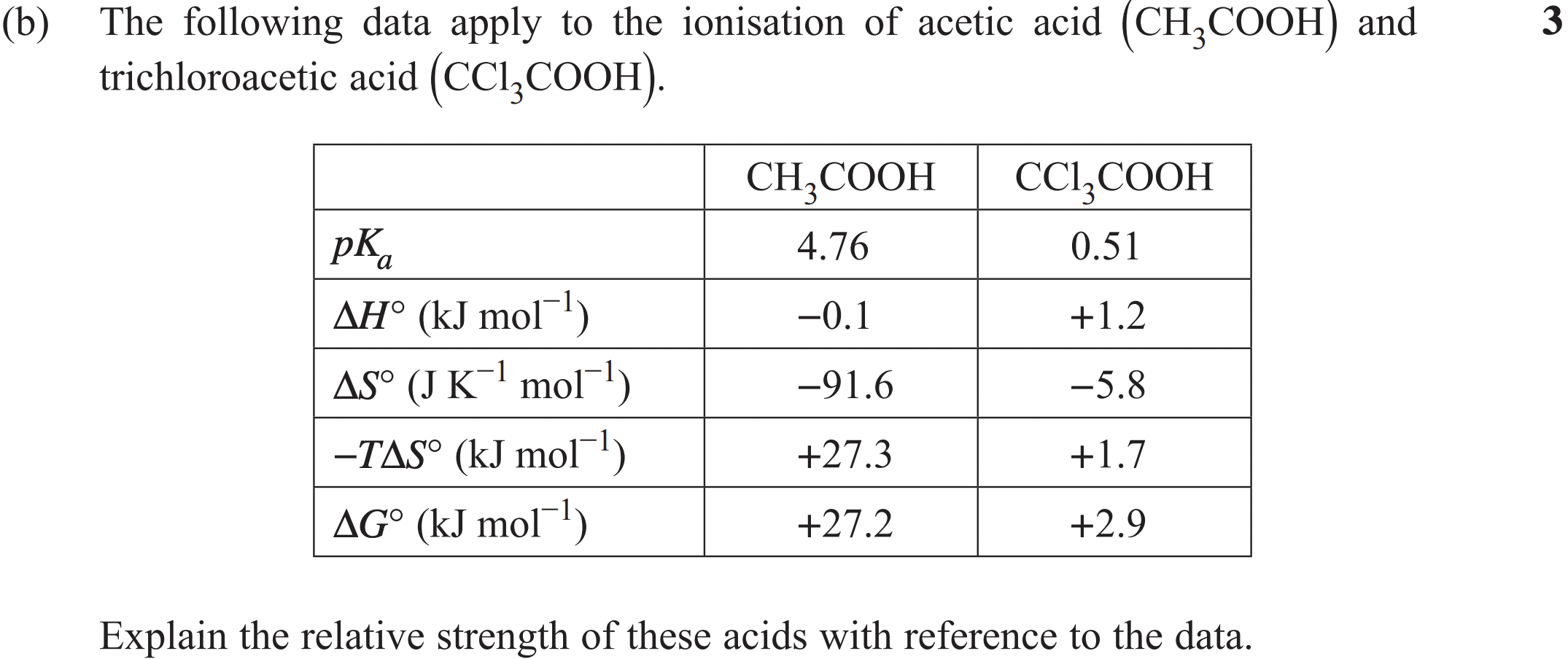
Trichloroacetic acid (CCl₃COOH) is the stronger acid because it has a smaller pKa and so, larger Ka. CCl₃COOH has a positive enthalpy which is not favourable and a negative entropy which is also not favourable. This leads to a positive Gibbs free energy which means its dissociation is non-spontaneous. While CCl₃COOH is the relatively stronger acid, it is not a strong acid that dissociates completely.
Acetic acid (CH₃COOH) has a favourable negative enthalpy but its entropy is much more unfavourable than CCl₃COOH (-91.6 compared to -5.8 J/K/mol). This leads to a much more positive Gibbs free energy, meaning the dissociation of acetic acid is even less spontaneous so it is the weaker acid.
Dilute H₂SO₄ is a hydrating agent. A must be an alkene that is hydrated into the alcohol B. KMnO₄ is an oxidising agent. Tertiary alcohols cannot oxidise so B must be a primary or secondary alcohol. C is either an aldehyde, carboxylic acid or ketone as these are the possible products of oxidising an alcohol.
The general formula for an alkene is CₙH₂ₙ. If the molar mass of A is 84.156 g/mol, the alkene must be hexene.
12.01 x n + 1.008 x 2n = 84.156
14.026n = 84.156
n = 84.156/14.026 = 6
The ¹³C NMR spectrum of A shows only 3 unique carbon environments, indicating a symmetrical structure that allows multiple carbons to have the same environment. The double bond must be between carbons 3 and 4. Compound A is 3-hexene.
Since 3-hexene is symmetrical, hydrating it will produce 3-hexanol which is Compound B. Markovnikov’s rule does not apply. This is confirmed by the infrared spectrum which shows a broad peak at 3400 cm⁻¹ for an OH group of an alcohol.
3-hexanol is a secondary alcohol and oxidising it will produce 3-hexanone, which is Compound C. The ¹H NMR spectrum of C has two peaks between 2.0 and 2.6 ppm but no peak between 9.4 to 10.0 ppm or 9.0 to 13.0 ppm. This confirms that Compound C must be a ketone and not an aldehyde or carboxylic acid. Because the double bond in the original alkene (Compound A) was not on a terminal carbon, an aldehyde or carboxylic acid cannot be produced anyway.
Compound A is 3-hexene, B is 3-hexanol and C is 3-hexanone.
Q = [CO₂]/[CO]² = (1.21 x 10⁻³)/(1.10 x 10⁻²)² = 10.0
Q = K so the system is at equilibrium.